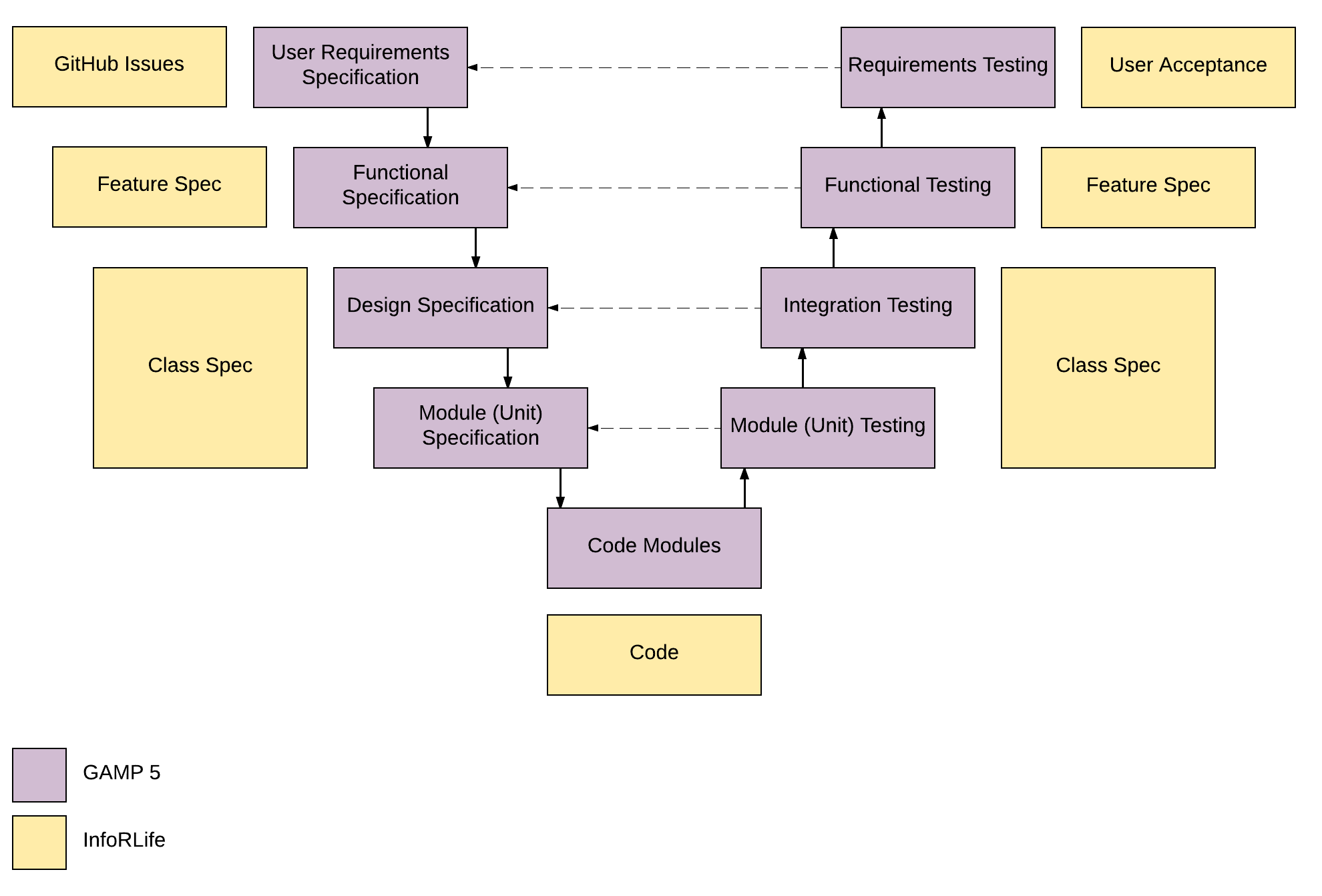
  Manufacturers need to define their approach to the life cycle management of the computerised systems responsible for producing the products they are making - all the way from concept and implementation through to operation and retirement.Īll of the above needs to be defined and documented within a Quality Management System, showing how system requirements are specified in the concept phase, how a system is to be released and maintained and then how it is to be retired. Owning an intimate knowledge of the product means manufacturers will be better placed to determine how potential changes to the system could affect the overall safety of the end product and the risk of harm to individuals.Īttention should, at all times, be focused on all those aspects of the system that are critical to ‘patient safety, product quality and data integrity’.ĭHF, DMR and DHR: Demystifying FDA medical device development requirements 2. They need this understanding to make critical science and risk-based decisions to ensure computerised manufacturing systems are ‘fit for use’.Īn understanding of product and process is also vital to minimising the risk of failure to systems within the operation cycle of software. #GAMP 5 SOFTWARE CATEGORIES PLUS#Manufacturers must understand exactly what the product is - its intended use and purpose - plus the purpose of the processes needed to develop it, in order to determine system requirements accurately. Five principles of GAMP 5’s risk-based approach to compliant GxP Computerised Systems 1. GAMP 5 ®, therefore, sets out guidelines for systems’ validation governed by five key concepts rooted in risk-based thinking. 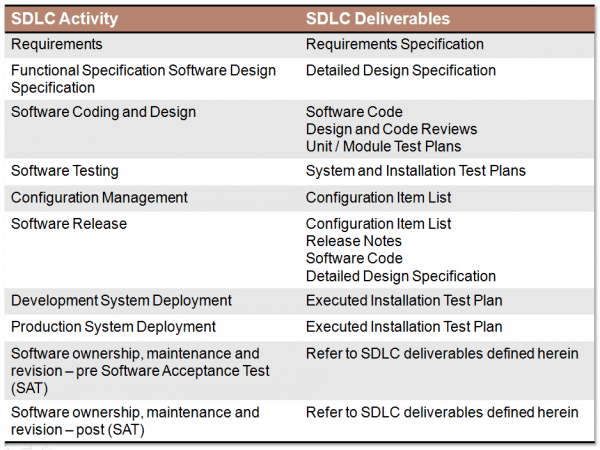
One of the central tenets of GAMP 5 ® is that quality cannot be tested into a batch of product or device, but instead needs to be built into every stage of the manufacturing process. ‘a cost effective framework… to ensure that computerised systems are fit for use and compliant with regulation’. Thus, the publication brings together the most up-to-date thinking on approaches to systems validation providing “a prescriptive method or standard, but rather provides pragmatic guidance, approaches and tools for the practitioner” GAMP 5 ® is the ISPE's best practice guidance which has been accepted by regulators worldwide (including the FDA) and is widely referenced in their literature and documentation.Īccording to the ISPE their publication does not represent GAMP is dedicated to creating global “communities of practice” in Pharmaceutical engineering and med tech. In doing so they detail a recognised standard for computer system validation (CSV) underpinned by the risk-based approach which is so much a feature of the regulatory landscape today. These guidelines are intended to ensure that pharmaceutical or medical device products are manufactured to the required quality. The Good Automated Manufacturing Practice (GAMP 5 ®) guidelines define a set of core principles for GxP, as produced by the ISPE (International Society for Pharmaceutical Engineering). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
